The animal-vegetal axis and mesoderm formation
A precondition to form the amphibian organizer is the formation of mesoderm at an equatorial zone between the animal and vegetal pole. Since axes formation, e.g., in a mouse is possible even in the absence of maternal determinants, a mechanisms is expected that allow the de novo generation of the animal-vegetal axis: a self-enhancing feedback loop combined with an inhibitory component of longer range must exist. A key gene for the endodermal pathway is T-box transcription factor VegT [1-3]. The mutual activation of VegT and derriere [4] and possibly other TGF-ß related signaling molecules satisfy the condition of autocatalysis. The evidence for the existence of a long-range inhibitory component is more circumstantial. This activation remains restricted to the vegetal pole despite the fact that the secretion of derriere should lead to a wave-like spread of the VegT / derriere activation. The animal cap assay demonstrates that this is not because the remaining cells are not competent. To the contrary, injection of VegT RNA together with a lineage tracer into a single animal cap cell at the 32-cell stage has revealed that only a fraction of the progenitor cells express that gene later, suggesting that the extension of the region in which the gene becomes activated is confined by an inhibitory reaction of longer range [2]. Therefore, all the ingredients predicted by our general model of pattern formation seem to be present. To initiate a self-enhancing reaction some basal activity is required. The maternally supplied VegT - RNA can fulfill this function. Its asymmetric deposition would be decisive as to where the maximum will arise. In the simulation shown below, the ectodermal pathway (blue) is assumed to be a default state. A pattern forming system generates a patch-like activation (red) causing endoderm formation and the suppression of the default state.
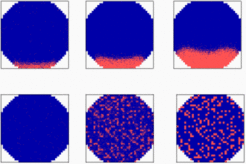
In the simulation shown above, the endoderm (red) forms by a pattern forming reaction. This gene activation is connected with a suppression of genes responsible for ectoderm formation (blue). In the absence of activator diffusion (community effect), the endoderm cells appear in an intermingled arrangement.
Separation of the germ layers: zones or individual cells?
In amphibians, well-separated zones with endo- and mesodermal specification, respectively, are formed before gastrulation begins. In the zebra fish, both cell types are more intermingled and the progeny of a single cell can give rise to both cell types, indicating that the pattern formation is still going on [5,6](Warga and Nüsslein-Volhard, 1999; Rodaway et al., 1999). Similarly, in the chick, individual cells separate from the epiblast and ingress to form meso- and endoderm. In some Cnidaria including hydra, the intermingled determination of ecto- and endoderm seems to be even more extreme. First a single layered coeloblastua is formed from which individual cells delaminate and ingress to form later the endoderm of the gastric cavity [7] . Although molecular data are not yet available, this behavior suggests that the choice between the two pathways is made first in a salt and pepper-like fashion in the single-layered cell sheet, followed by an individual ingression of the endodermal cells.
According to the model, whether differentiated cells emerge as coherent regions in zones or individually on a cell-by-cell basis depends on the following factors. If the self-enhancing reaction can spread, large coherent patches are expected (community effect). If, e.g., by maternal determinants or preceding pattern forming reactions a preferential initiation site exists, such a zone appear at predictable positions. Alternatively, if the feedback loop does not involve secreted molecules or if their spread is limited, the decision is made on a cell-by-cell basis and random fluctuations can be decisive. Again, a condition for the salt and pepper pattern is a saturation of the autocatalysis, otherwise each cell will be surrounded by a halo of inhibition. All intermediate patterns between zones and salt-and-pepper distributions are possible under appropriate conditions. The ratio at which different cell types emerge is regulated.
Mesoderm formation

Simulation of mesoderm formation: the endoderm acts as a local source of a long ranging signal that activates the feedback loop responsible for mesoderm formation (green).
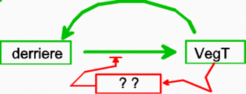
High activity of VegT not only gives rise to the endoderm but also induces ectodermal cells to form mesoderm at the zone of juxtaposition. An activator-inhibitor system has been identified involved in this process: Nodal / lefty2. Nodal activation has a self-enhancing component. It also regulates the production of its antagonist, Lefty2, which, in turn, blocks nodal activation [8,9](Bisgrove et al., 1999; Chen and Schier, 2002; Branford and Yost, 2002). The transcription factor Brachyury is a pan-mesodermal marker in vertebrates. Its activation depends on a positive feedback involving a secreted signaling substance FGF [10]. TGF-ß related factors such as Nodal are candidates for the activation of this mesoderm-defining loop [2,3]. Thus, although for instance, both Xnr-1 and eFGF are secreted factors that activate the pan-mesodermal marker Brachyury, in terms of the model, both have non-redundant functions. The first causes the long-range activation by the endoderm, the latter is involved in its maintenance through a mesoderm-specific feedback loop. As expected, an antagonistic component should be involved that restricts this autoregulatory loop (antivin ; Cheng et al., 2000). Therefore, known molecular components in suggest that mesoderm formation is based on a process with pattern capability.
Further Reading and References
Meinhardt, H. (2001). Organizer and axes formation as a self-organizing process. Int. J. Dev. Biol. 45, 177-188 [PDF]
- Zhang, J. and King, M.L. (1996). Xenopus VegT RNA is localized to the vegetal corte during oogenesis and encodes a novel T-box transcription factor involved in mesodermal patterning. Development 122, 4119-4129.
- Clements, D., Friday, R.V. and Woodland, H.R. (1999). Mode of action of VegT in mesoderm and endoderm formation. Development 126, 4903-4911.
- Kofron, M., Demel, T., Xanthos, J., Lohr, J., Sun, B., Sive, H., Osada, S., Wright, C., Wylie, C. and Heasman, J. (1999). Mesoderm induction in Xenopus is a zygotic event regulated by maternal vegt via tgf beta growth factors. Development 126, 5759-5770.
- Sun, B.I., Bush, S.M., Collins-Racie, L.A., LaVallie, E.R., DiBlasio-Smith, E.A., Wolfman, N.M., McCoy, J.M. and Sive, H.L. (1999). derriere: a TGF-beta family member required for posterior development in Xenopus. Development 126, 1467-1482.
- Warga, R.M. and Nüsslein-Volhard, C. (1999). Origin and development of the zebrafish endoderm. Development 126, 827-838.
- Rodaway, A., Takeda, H., Koshida, S., Broadbent, J., Price, B., Smith, J.C., Patient, R. and Holder, N. (1999). Induction of the mesendoderm in the zebrafish germ ring by yolk cell-derived TGF-beta family signals and discrimination of mesoderm and endoderm by FGF. Development 126, 3067-3078.
- Martin, V.J., Littlefield, C.L., Archer, W.E. and Bode, H.R. (1997). Embryogenesis in hydra. Biological Bulletin 192, 345-363.
- Chen, Y. and Schier, A.F. (2002). Lefty proteins are long-range inhibitors of squint-mediated nodal signaling. Curr Biol 12, 2124-2128.
- Branford, W.W. and Yost, H.J. (2002). Lefty-dependent inhibition of nodal- and wnt-responsive organizer is essential for normal gene expression gastrulation. Curr Biol 12, 2136-2141.
- Schulte-Merker, S. and Smith, J.C. (1995). Mesoderm formation in response to brachyury requires FGF signaling. Current Biol. 5, 62-67.
- Cheng, A.M.S., Thisse, B., Thisse, C. and Wright, C.V.E. (2000). The lefty-related factor xatv acts as a feedback inhibitor of nodal signaling in mesoderm induction and l-r axis development in Xenopus. Development 127, 1049-1061.


